Master healthcare equipment procurement. Learn regulatory requirements, GPO considerations, and spec comparison for medical buyers.
Healthcare equipment procurement operates under unique constraints. Regulatory requirements, clinical considerations, patient safety, and complex stakeholder dynamics make medical equipment purchases fundamentally different from other procurement.
This comprehensive guide covers what healthcare buyers need to know when evaluating and purchasing medical equipment—from regulatory fundamentals through vendor selection and ongoing management.
Why Healthcare Procurement Is Different
Regulatory Complexity
| Regulatory Body | Jurisdiction | Key Requirements |
|---|
| FDA | US Federal | Device classification, 510(k)/PMA clearance |
| State health departments | State | Licensing, reporting |
| The Joint Commission | Accreditation | Standards, survey compliance |
| CMS | Federal reimbursement | Conditions of participation |
| OSHA | Worker safety | Radiation, infection control |
Critical: Every procurement decision has regulatory implications. Non-compliance risks patient safety, accreditation, and legal liability.
Patient Safety Focus
| Safety Consideration | Procurement Impact |
|---|
| Clinical outcomes | Equipment affects treatment success |
| Infection risk | Design affects cleaning, sterilization |
| User error | Interface design affects mistakes |
| Equipment failure | Reliability is critical for life-support |
| Electromagnetic compatibility | Must not interfere with other devices |
Clinical Stakeholder Complexity
| Stakeholder | Primary Concerns |
|---|
| Physicians | Clinical capability, outcomes, workflow |
| Nurses | Daily usability, patient care, workload |
| Clinical engineering | Maintenance, support, reliability |
| IT | Integration, security, data |
| Finance | Cost, reimbursement, ROI |
| Infection control | Cleaning, sterilization |
| Risk management | Liability, safety, adverse events |
Regulatory Fundamentals
FDA Device Classification
| Class | Risk Level | Regulatory Pathway | Examples |
|---|
| Class I | Lowest | General controls, most exempt | Bandages, exam gloves |
| Class II | Moderate | 510(k) clearance | Ultrasound, infusion pumps |
| Class III | Highest | PMA approval | Pacemakers, high-risk implants |
510(k) Verification
| Element | What to Verify |
|---|
| 510(k) number | Valid FDA clearance number |
| Cleared indications | Matches your intended use |
| Substantial equivalence | Based on legally marketed predicate |
| Listing status | Current FDA registration |
Pro Tip: Verify independently—search FDA databases. Don't rely solely on vendor claims.
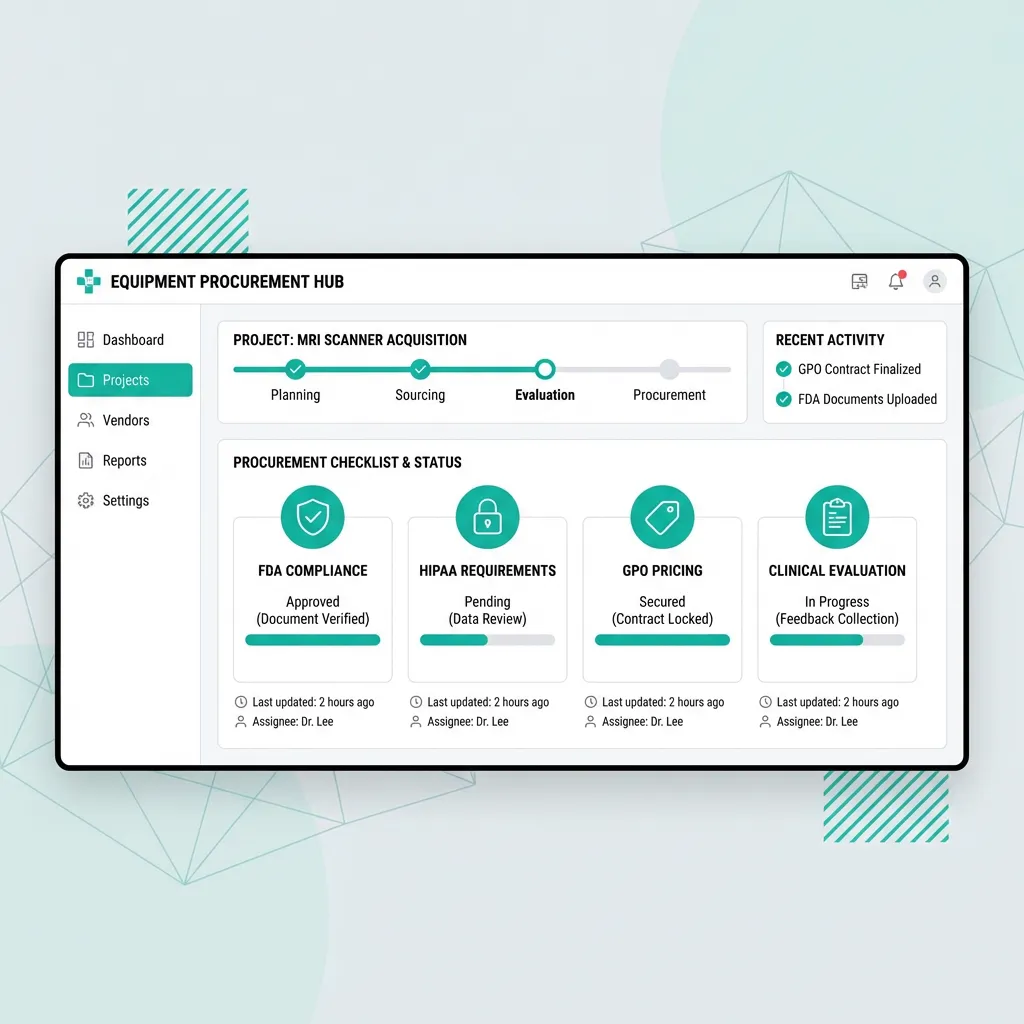
GPO and Purchasing Programs
Understanding GPOs
| GPO Benefit | Description |
|---|
| Contract pricing | Pre-negotiated discounts |
| Contract convenience | Already negotiated terms |
| Vendor vetting | Qualification already completed |
| Compliance support | Pricing transparency |
Major GPOs: Premier, Vizient, HealthTrust, Intalere, GNYHA
When to Use GPO vs. Direct
| Use GPO When | Go Direct/RFP When |
|---|
| Standard equipment | Highly customized needs |
| Time-constrained | Unique requirements |
| Staff-limited | Volume justifies negotiation |
| Commodity items | Strategic partnerships |
Clinical Evaluation Process
Building the Evaluation Team
| Team Member | Role in Evaluation |
|---|
| Clinical end user | Workflow, clinical needs, usability |
| Clinical engineering | Technical assessment, service needs |
| IT | Integration, security, data |
| Infection control | Cleaning requirements, safety |
| Supply chain | Procurement process, sourcing |
| Finance | Cost analysis, ROI |
Clinical Trials and Demonstrations
| Evaluation Type | Purpose | Duration |
|---|
| Demonstration | See equipment in action | Hours |
| Hands-on evaluation | Users test equipment | Days |
| Clinical trial | Use with patients | Weeks |
| Extended trial | Full workflow integration | Months |
Medical Equipment Specifications
Imaging Equipment
| Specification | What to Evaluate |
|---|
| Image resolution | Spatial, contrast resolution |
| Scan time | Throughput capacity |
| Dose management | Radiation exposure (CT, X-ray) |
| Field of view | Coverage area |
| Patient accommodation | Weight limit, bore size |
| DICOM compliance | Image format, connectivity |
Patient Monitoring
| Specification | What to Evaluate |
|---|
| Parameters | What's measured |
| Alarm management | Customization, escalation |
| Battery life | Portable use duration |
| Interface | Ease of use, visibility |
| Integration | EHR connectivity, central station |
Infusion Devices
| Specification | What to Evaluate |
|---|
| Flow rates | Range and accuracy |
| Drug library | Pre-programmed medications |
| Safety features | Dose error reduction |
| Interoperability | EHR integration, barcode |
Cybersecurity in Medical Devices (IoMT)
⚠️ IoMT Security Requirements
- MDS2 Form: Demand the Manufacturer Disclosure Statement for Medical Device Security
- Patch Management: Vendor must commit to security patches for device's service life
- Data Governance: Where does patient data go? Cloud? On-prem? Who owns it?
Total Cost Analysis
| Cost Component | Healthcare Specifics |
|---|
| Equipment purchase | List price, GPO pricing, negotiated |
| Installation | Medical gas, electrical, structural |
| Training | Clinical staff, multiple shifts |
| Consumables | Supplies, accessories, reagents |
| Service | Warranty, service contracts |
| Integration | IT, EHR connectivity |
Vendor Evaluation
| Criterion | What to Evaluate |
|---|
| Regulatory history | FDA warning letters, recalls |
| Clinical outcomes | Published evidence |
| Customer satisfaction | KLAS, MD Buyline ratings |
| Service capability | Local support, response |
| Financial stability | Long-term vendor viability |
Independent Sources
| Source | Type of Information |
|---|
| KLAS Research | Customer ratings, performance |
| ECRI | Technology assessments, safety |
| FDA MAUDE | Adverse event reports |
| MD Buyline | User ratings, purchasing data |
Healthcare Procurement Checklist
Regulatory Verification
- ☐ FDA clearance verified (510(k)/PMA)
- ☐ Device classification confirmed
- ☐ Intended use matches clearance
- ☐ UDI compliance checked
- ☐ State requirements reviewed
Clinical Evaluation
- ☐ Evaluation team assembled
- ☐ Demonstration conducted
- ☐ Clinical trial completed (if applicable)
- ☐ User feedback collected
- ☐ Clinical outcomes reviewed
Vendor Due Diligence
- ☐ Regulatory history checked
- ☐ References verified
- ☐ Independent ratings reviewed
- ☐ Financial stability confirmed
- ☐ Contract terms negotiated
Procure with Confidence
Healthcare equipment procurement is complex but manageable with structured approach. Systematic specification comparison combined with clinical evaluation and regulatory diligence leads to decisions that serve patients, clinicians, and the organization.
See Healthcare Solutions → | Vendor Risk Assessment →
Ready to Transform Your Procurement Process?
Try SpecLens today and experience AI-powered specification comparison. Save time, reduce errors, and make better purchasing decisions.


